
The pi electrons are not as fully under the control of the carbon nuclei as the electrons in the sigma bond and, because they lie exposed above and below the rest of the molecule, they are relatively open to attack by other things. 
The electrons in the pi bond are free to move around anywhere in this shaded region and can move freely from one half to the other. The other pair of electrons is found somewhere in the shaded part above and below the plane of the molecule. A pi bond, let me go ahead and write that here. We get side by side overlap of our P orbitals and this creates a pi bond. The sigma bond between to the carbon atoms is formed from overlap of sp hybrid orbitals from each. These unhybridized P orbitals can overlap side by side. The triple bond is composed of one bond and two bonds. Note that sigma bond has been referred to as the strongest type of covalent bond because the extent of overlap is maximum in case of orbitals involved in the formation of the sigma bond. In Sigma bond the electron density is maximum and it is cylindrically symmetrical about the bond axis. A total of five sigma bonds and then we have a new type of bonding. (b) shows hydrogens antibonding orbital, which is higher in energy and is normally not occupied by any electrons. In organic chemistry, hyperconjugation ( -conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily -character. In (a) the two nuclei are surrounded by a cloud of two electrons in the bonding orbital that holds the molecule together. This sort of bond is called a sigma bond. Sigma bonds (\sigma) () are the strongest type of covalent bond, formed by head-on overlapping of atomic orbitals. Covalent bonding of two hydrogen atoms to form a hydrogen molecule, H. In this diagram, the line between the two carbon atoms represents a normal bond - the pair of shared electrons lies in a molecular orbital on the line between the two nuclei where you would expect them to be. A molecular orbital is a region of space within the molecule where there is a high probability of finding a particular pair of electrons. 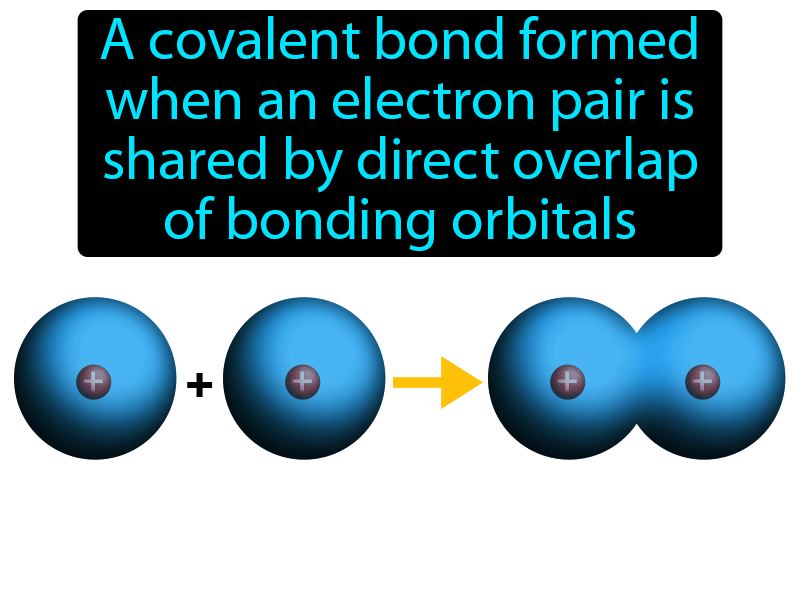
One of the pairs of electrons is held on the line between the two carbon nuclei as you would expect, but the other is held in a molecular orbital above and below the plane of the molecule. What the diagram doesn't show is that the two pairs aren't the same as each other. Explanation: Here we discuss about Lewis structure, sigma and pi bonds. The double bond between the carbon atoms is, of course, two pairs of shared electrons. What is true of C=C in ethene will be equally true of C=C in more complicated alkenes.Įthene, C 2H 4, is often modeled as shown on the right. We are going to start by looking at ethene, because it is the simplest molecule containing a carbon-carbon double bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
